 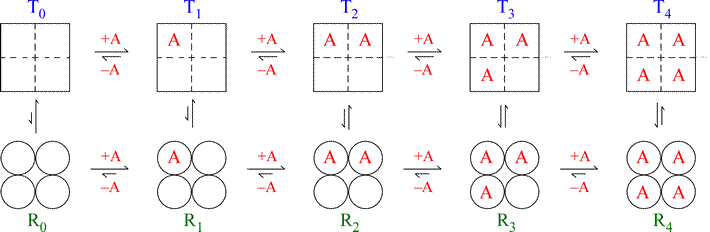 
This process is experimental and the keywords may be updated as the learning algorithm improves. These keywords were added by machine and not by the authors. When multiple enzymes work in concert for some cellular process, and are regulated in concert by the same original effector, this may produce very high ultrasensitivity. The H-bond modeling observations therefore strongly support the biochemical observation of positive cooperativity with a Hill coefficient of 1.52, supporting a robust cooperativity between non. This controlled change in affinity makes the enzyme more sensitive to changes in substrate concentration (positive cooperativity), or dampens the enzyme’s response to changes in substrate concentration (negative cooperativity). Most K-type enzymes have positive cooperativity a limited number of regulatory enzymes have negative cooperativity. This is also true in the conversion of the R state to the T state- all bounded oxygen must be released before a full conversion can take place.The MWC model postulates that all subunits in an enzyme oligomer change conformation in a concerted fashion, while the KNF model assumes this change may be sequential for the different subunits. The biochemical reaction kinetics of those enzymes which have single active site follows Michaelis-Menten model. In the concerted model, all oxygen binding sites on Hemoglobin in the T state must be filled before the molecule converts to the R state. Hill’s Coefficient Cooperativity Positive Cooperativity o The Concerted model o The sequential Model Negative Cooperativity. When the model has high oxygen affinity, it means that it is highly R state favored and hemoglobin is T state favored when it has no bounded oxygen. Equilibrium is shifted between both states. The T-state is the deoxy form of hemoglobin and the R-State is the fully oxygenated form. Molecules can exist either in the T (tense) state, or R (relaxed) state. In a real system, properties from both models are needed to explain the behavior of hemoglobin. This model and the sequential model displays the extreme cases of R and T transitions. Allosteric effectors of hemoglobin, such as 2,3-BPG, function by shifting the equilibrium towards or away from the T-state, depends on whether it's an inhibitor or a promoter. This means that at high oxygen levels, the R form will be prevalent and at lower oxygen levels, the T form will be prevalent. Overall, oxygen binding shifts the equilibrium toward the R state. Two models are often used to explain cooperativity in multimeric proteins: the concerted and the sequential model. Thus in the concerted model of the hemoglobin, it shows that the one oxygen binding to an active site will increase the probability of other oxygen binding to the other active sites in the hemoglobin. The binding of oxygen at one site increases the binding affinity in other active sites. can account for positive cooperativity, while the KNF. The difference in deoxyhemoglobin and oxyhemoglobin is that the "deoxy" form doesn't have Oxygen and the "oxy" form is highly oxygen bounded. sequential model of Koshland, Nemethy and Filmer were formulated to account for positively co. The sequential model can also account for negative homotropic effects, or negative cooperativity, whereas the allosteric model cannot. The ESI-MS data are consistent with a binding model, proposed by Homans and coworkers, in which ligand binding to CTB5 can be described by three intrinsic Ka. T state is constrained due to the subunit-subunit interactions while the R state is more flexible due to the ability of oxygen binding. For positive cooperativity they predict binding curves that are so similar that it is very difficult to distinguish experimentally between them. 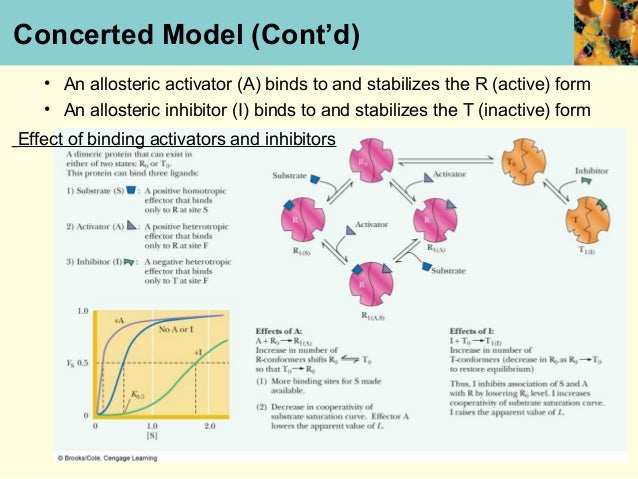
The T state of the hemoglobin is more tense as it is in the deoxyhemoglobin form while the R state of the hemoglobin is more relaxed as it is in the oxyhemoglobin form. It focuses on the two states of the Hemoglobin the T and R states. The diagram illustrates the 'sequential' model of cooperativity, which suggests that one subunit of haemoglobin starts a sequence of conformational changes in the other subunits haemoglobin which increase their affinity for. The Concerted Model, also known as MWC model or symmetry model, of hemoglobin is used to explain the cooperativity in oxygen binding as well as the transitions of proteins which made up of identical subunits. The p50 value, positive cooperativity and the T / R states explaining the curve were all rewarded with extra marks. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
